|
We found that the proportion of patients receiving chemotherapy decreased with age 80.0%, 70.4%, 50.6%, and 30.2% of patients aged 70–74, 75–79, 80–84, and ≥ 85 years, respectively, received chemotherapy. 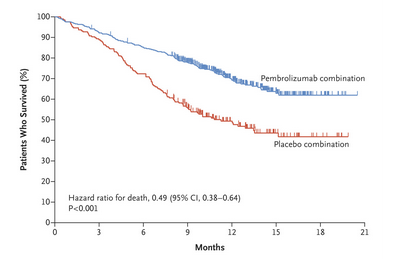
The first-line chemotherapy regimens among 9,737 patients who were diagnosed with advanced lung cancer between January and December 2013, were identified and compared based on age. Data from the survey database of Diagnostic Procedure Combination and hospital-based cancer registries of designated cancer centers nationwide were used. This study aimed to examine the prescribing patterns of first-line chemotherapy according to age in the real-world practice. In clinical practice, physicians are required to decide the treatment based on a lack of enough evidence. In Japan, single agent chemotherapy was recommended for advanced non-small cell lung cancer (NSCLC) for those, who were aged ≥75 years, while the Western guidelines did not recommend a specific regimen. Incidence of grade 3-5 adverse events was similar in the pembrolizumab-combination (71.9%) and placebo-combination (66.8%) groups.įirst-line pembrolizumab plus pemetrexed-platinum continued to demonstrate substantially improved OS and PFS in metastatic nonsquamous NSCLC, regardless of PD-L1 expression or liver/brain metastases, with manageable safety and tolerability.Aging of the population has led to an increase in the prevalence of cancer among older adults. OS and PFS benefits with pembrolizumab were observed regardless of PD-L1 expression or presence of liver/brain metastases. Median (95% CI) time from randomization to objective tumor progression on next-line treatment or death from any cause, whichever occurred first (progression-free-survival-2 PFS-2) was 17.0 (15.1 to 19.4) months and 9.0 (7.6 to 10.4) months, respectively (HR, 0.49 95% CI, 0.40 to 0.59). No alpha was assigned to this updated analysis.Īs of Septem(median follow-up, 23.1 months), the updated median (95% CI) OS was 22.0 (19.5 to 25.2) months in the pembrolizumab-combination group versus 10.7 (8.7 to 13.6) months in the placebo-combination group (hazard ratio, 0.56 95% CI, 0.45 to 0.70]). Response was assessed per RECIST (version 1.1) by central review. Eligible patients with disease progression in the placebo-combination group could cross over to pembrolizumab monotherapy. Patients were randomly assigned (2:1) to receive pemetrexed and platinum plus pembrolizumab (n = 410) or placebo (n = 206) every 3 weeks for 4 cycles, then pemetrexed maintenance plus pembrolizumab or placebo for up to a total of 35 cycles. We report an updated analysis from KEYNOTE-189 (: NCT02578680). In KEYNOTE-189, first-line pembrolizumab plus pemetrexed-platinum significantly improved overall survival (OS) and progression-free survival (PFS) compared with placebo plus pemetrexed-platinum in patients with metastatic nonsquamous non‒small-cell lung cancer (NSCLC), irrespective of tumor programmed death-ligand 1 (PD-L1) expression. 24 Fondazione Istituto di Ricovero e Cura a Carattere Scientifico Istituto Nazionale dei Tumori, Milan, Italy.21 Kansai Medical University Hospital, Osaka, Japan.20 Chris O'Brien Lifehouse, Camperdown, New South Wales, Australia.19 Hospital Universitario Quirónsalud Madrid, Madrid, Spain.18 Department of Oncology, University of Turin, Azienda Ospedaliero-Universitaria San Luigi, Orbassano, Italy.17 David Geffen School of Medicine at University of California Los Angeles, Los Angeles, CA.16 LungenClinic, Airway Research Center North, German Center for Lung Research, Grosshansdorf, Germany.15 Epworth Healthcare, Richmond, Victoria, Australia.14 Fondazione IRCCS Cà Granda Ospedale Maggiore Policlinico, Milan, Italy.13 Soroka Medical Center, Ben-Gurion University, Beer Sheva, Israel.11 Sunnybrook Health Sciences Centre, Toronto, Ontario, Canada.9 Southern Medical Day Care Centre, Wollongong, New South Wales, Australia. 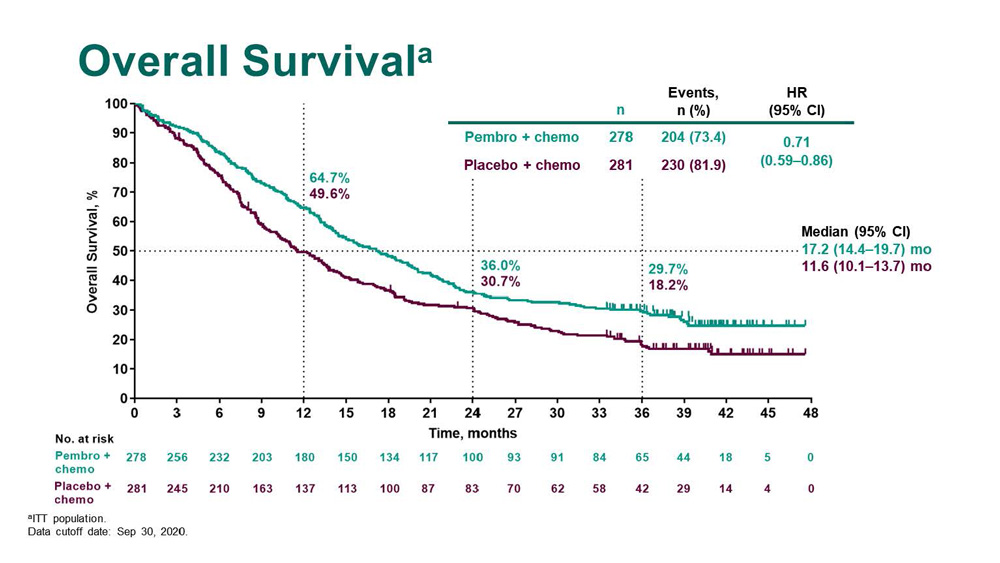 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
